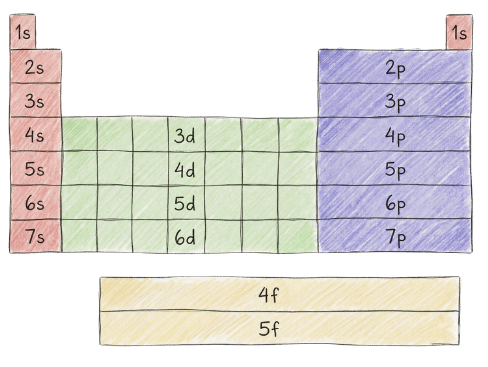
Electron Configuration
Core Concept
Definition: Electron configuration describes the distribution of electrons in an atom's orbitals, following specific rules.
Purpose: It explains an atom's chemical properties, periodic trends, and bonding behavior.
Electrons fill orbitals in order of increasing energy, and each subshell has a fixed capacity.
Valence electrons determine chemical properties and periodic trends.
Exceptions to configurations occur to achieve more stable half-filled or fully filled subshells.
Test Yourself
Related Examples and Practice Problems
Topic Overview Podcast
Topic Related Resources
|
LABORATORY
|
DEMONSTRATIONS
|
ACTIVITIES
|
VIRTUAL SIMULATIONS
|
3 Filling Rules for Electron Configuration
RULE #1: Aufbau Principle
Rule: Electrons fill the lowest energy orbitals first
Filling Order: 1s → 2s → 2p → 3s → 3p → 4s → 3d → 4p → 5s → 4d → 5p…
Key Point: 4s fills before 3d (4s is lower energy due to penetration)
RULE #2. Pauli Exclusion Principle
Rule: Maximum of 2 electrons per orbital
Requirement: If 2 electrons in same orbital, they must have opposite spins
Representation: ↑↓ (opposite arrows)
RULE #3. Hund's Rule
Rule: Half-fill all orbitals of same energy before pairing electrons
Example: 2p orbitals fill as [↑][↑][↑] before [↑↓][↑][↑]
Reason: Electrons prefer to be unpaired when possible
Notation
General Format:
n: Principal energy level.
l: Subshell type (s,p,d,fs, p, d, fs,p,d,f).
Superscript: Number of electrons in the subshell.
Example: $1s^2$ indicates 2 electrons in the 1s-orbital.
Shorthand Notation:
Use the previous noble gas in brackets to simplify the configuration.
Example: Sodium (Z=11):
Full: $1s^2 2s^2 2p^6 3s^1$
Shorthand: [Ne]$3s^1$
Exceptions to Electron Configuration
Some elements have electron configurations that deviate from the expected order to achieve more stable arrangements:
Chromium (Z=24):
Expected: $[\text{Ar}] 4s^2 3d^4$
Actual: $[\text{Ar}] 4s^1 3d^5$ (half-filled d-subshell is more stable).
Copper (Z=29):
Expected: $[\text{Ar}] 4s^2 3d^9$
Actual: $[\text{Ar}] 4s^1 3d^{10}$ (fully filled d-subshell is more stable).